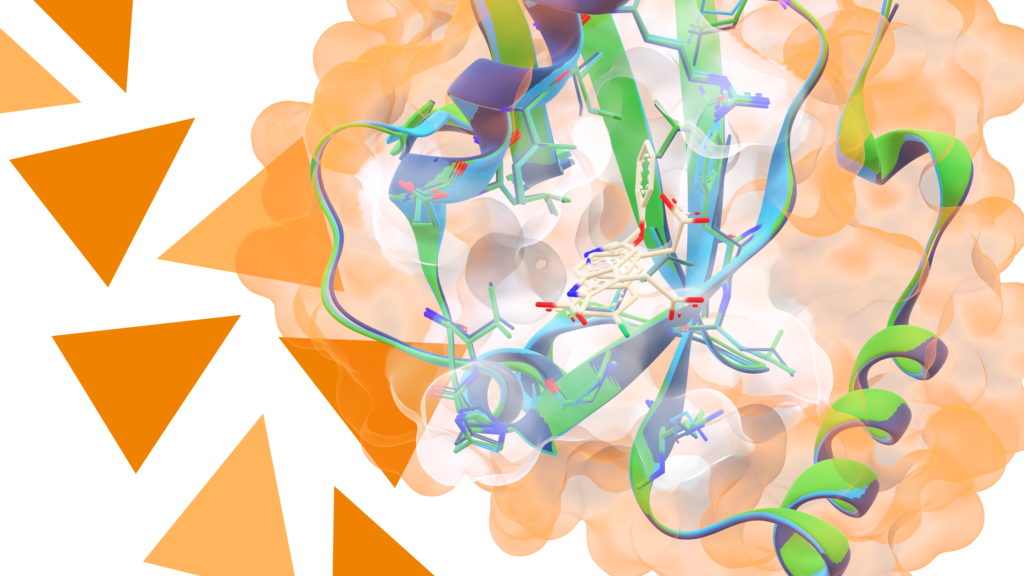
The challenges associated with anti-infective drug-discovery programmes can be tackled by combining antibacterial screening with several established and unprecedented hit-identification strategies in parallel. This approach will be illustrated using two targets: The first is the enzyme DXS from the methyl erythritol phosphate pathway, which is essential for pathogens such as Mycobacterium tuberculosis and Plasmodium falciparum for the biosynthesis of the essential isoprenoid precursors, but absent in humans. We identified inhibitors with promising in vitro and anti-infective activity (M. tuberculosis and P. falciparum) using a number of hit-identification strategies, namely ligand- and structure-based virtual screening, phage display, dynamic combinatorial chemistry and de novo fragment-based design.
The second target is a vitamin transporter from the energy-coupling factor (ECF) class, which consist of an energizing module and a substrate-binding protein (S-component). Different S-components can interact with the same energizing module. We designed and synthesized substrate analogues with high affinity (Kd = 4–660 nM) and confirmed the predicted binding mode. A structure-based virtual screening campaign provided us with the first allosteric inhibitors of the transporter for folate, which are not cytotoxic and show promising antibacterial (S. aureus and S. pneumoniae) activity.