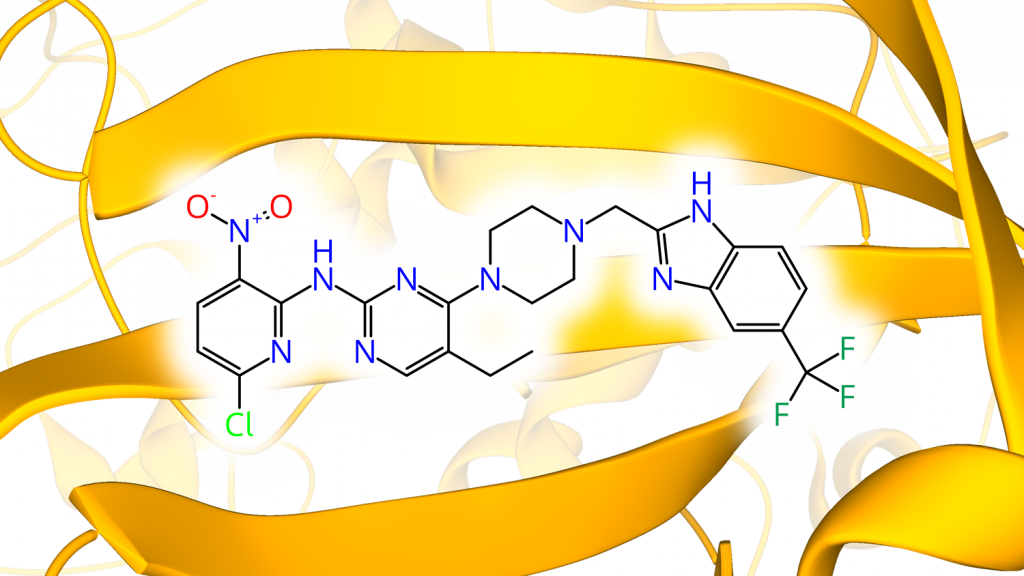
Protein kinases are now among the major drug targets with over 65 inhibitors approved since the groundbreaking approval of imatinib (Gleevec) in 2001. Covalent targeting approaches that have gained traction in recent years, have proven particularly useful in the protein kinase field. Typically, covalent inhibitors feature superior potency and efficacy as well as a prolonged duration of action. Of the >500 human protein kinases, the so-called “kinome”, almost 50% are characterized by the presence of an accessible, non-conserved cysteine residue in or around the ATP binding site. Addressing such cysteines by mild electrophiles, termed covalent “warheads”, enables the rational design of highly potent and selective protein kinase inhibitors as chemical probes or drugs. So far, however, only a small fraction of the protein kinases’ “cysteinome” has been addressed. In this talk, Matthias will present recent efforts toward the development of covalent inhibitors for protein kinases with a cysteine in the so-called “hinge region” using canonical and non-canonical warhead chemistry. Specifically, he will focus on inhibitors targeting TTK/MPS1, a kinase controlling the spindle assembly checkpoint, and S6K2/p70S6Kβ, a neglected member of the ribosomal protein S6 kinase family. Join us!