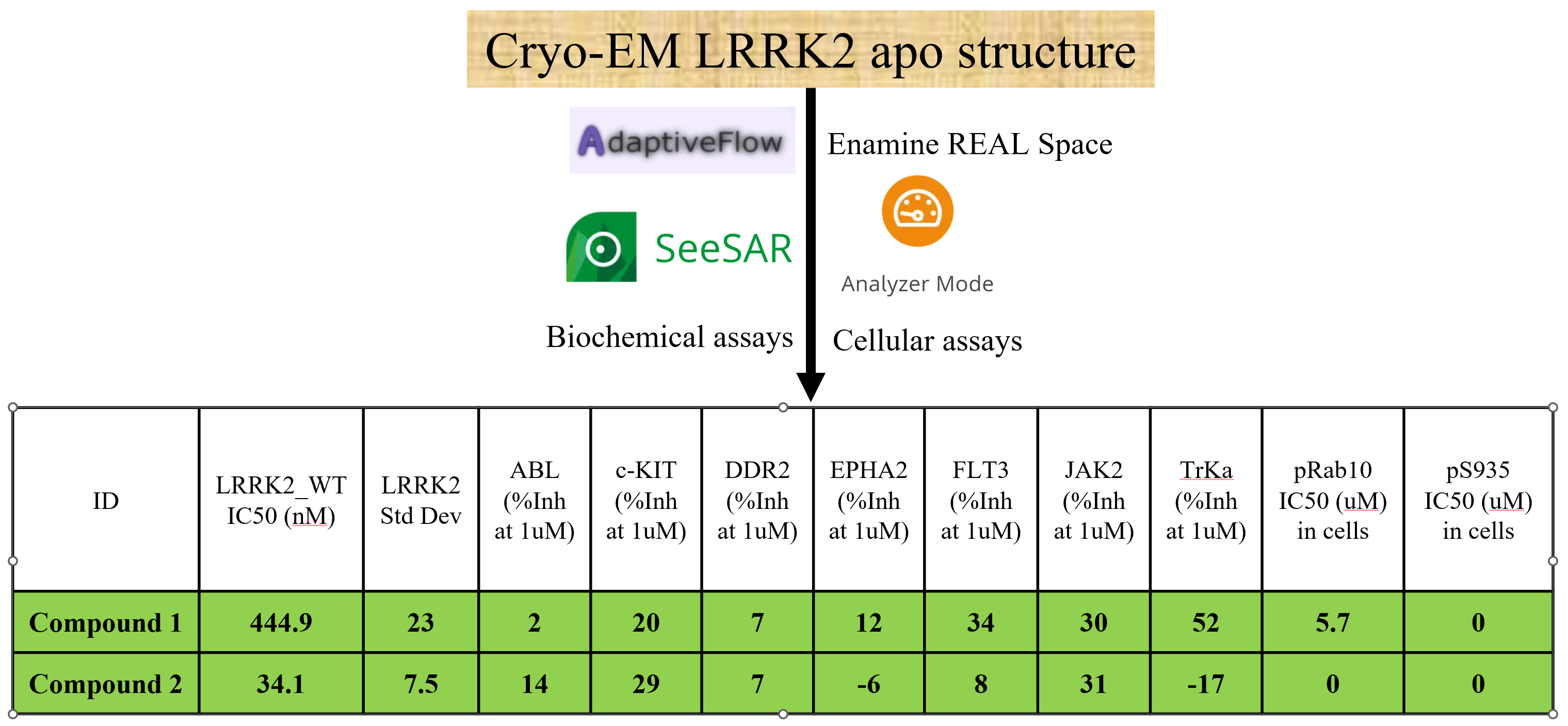
Structure-based virtual screenings targeting two allosteric sites in the LRRK2 structure characterized by Cryo-EM were performed, using in-house AdaptiveFlow open-source drug-discovery platform. Molecular filters were applied to the top hit candidates to only keep compounds with desirable properties.
All compounds were ordered from Enamine and tested by the in vitro biochemical assay, and by the ASMS direct binding assay. Top hits from these two assays were tested with the biochemical assay against a small kinase panel for their selectivity profile, as well as in cellular assays for their inhibition effect on LRRK2’s catalytic activity in cells. Compound 1 and 2 were confirmed to directly bind LRRK2 with a good selectivity across the tested small kinase panel. Notably, it remains to be confirmed by Cryo-EM that the two compounds bind to the docked allosteric sites of LRRK2.
After 3 months, Ming has achieved the following milestones:
- Identified two nM-range hits that directly bind to LRRK2 and have good selectivity, as confirmed by multiple in vitro assays and cellular functional assays.
- The more potent isomer of the more potent hit was determined in in vitro assays, and will be used for Cryo-EM experiments to characterize the LRRK2-inhibitor complex structure.
- To be Achieved with SeeSAR and InfiniSee