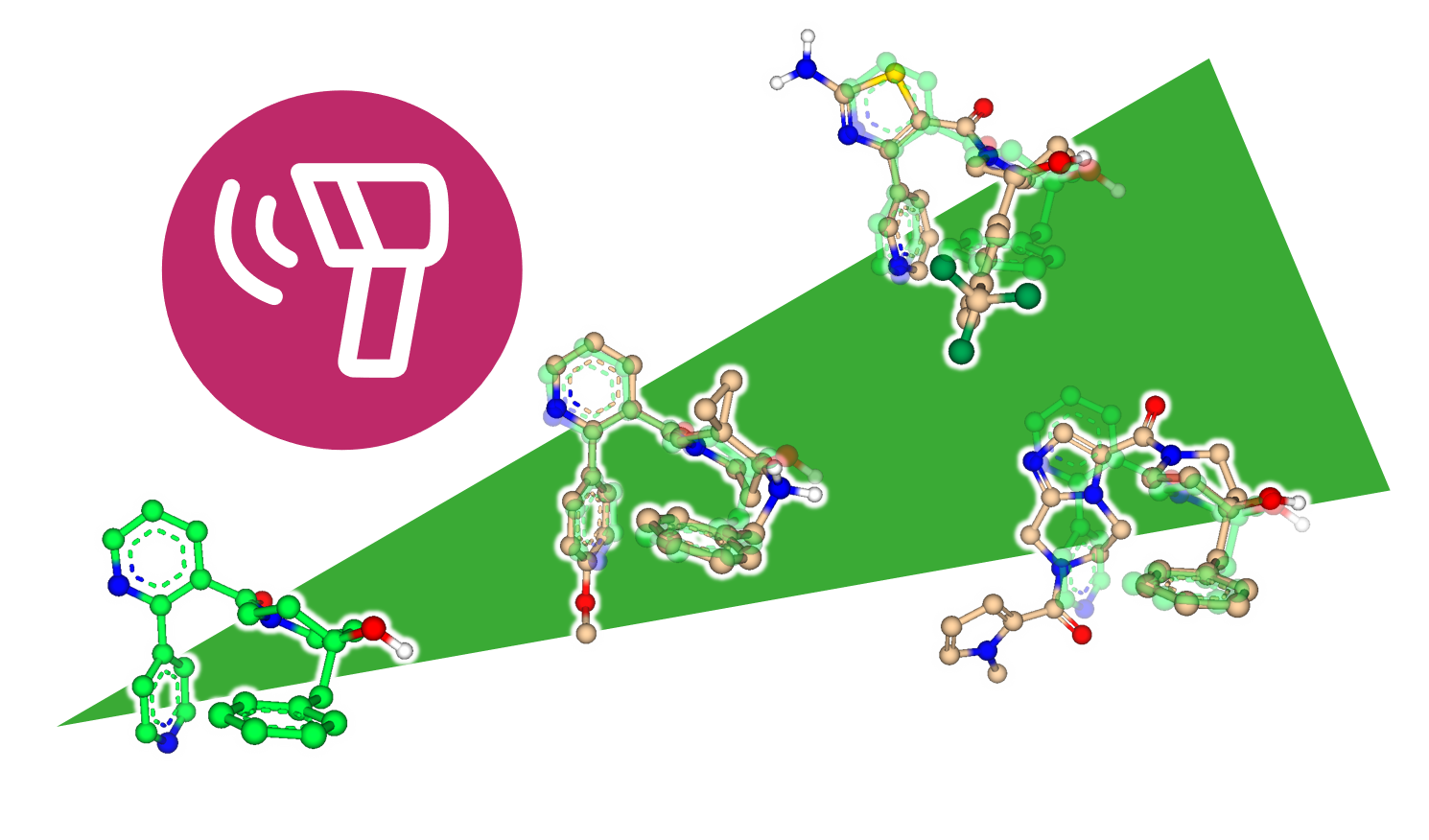
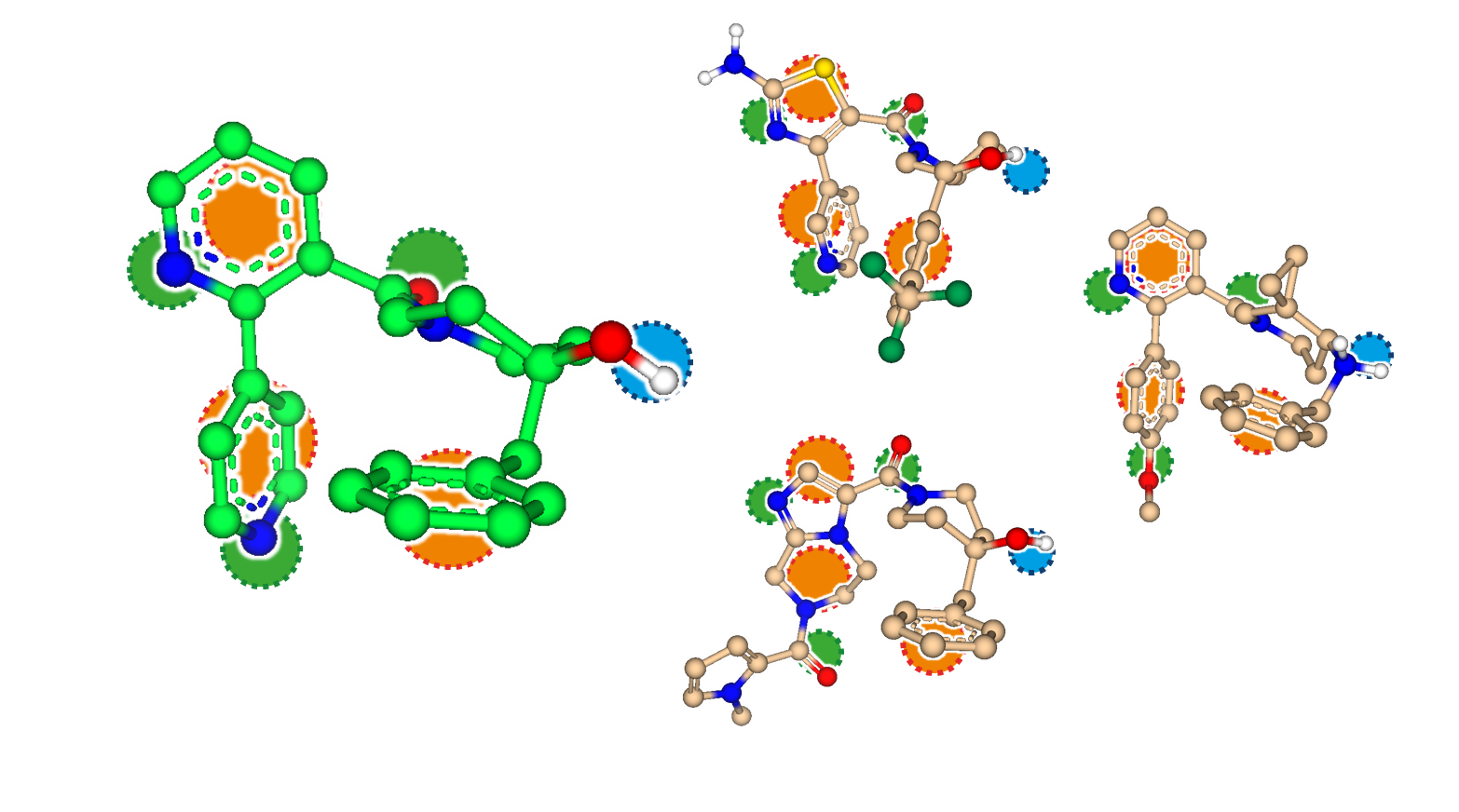
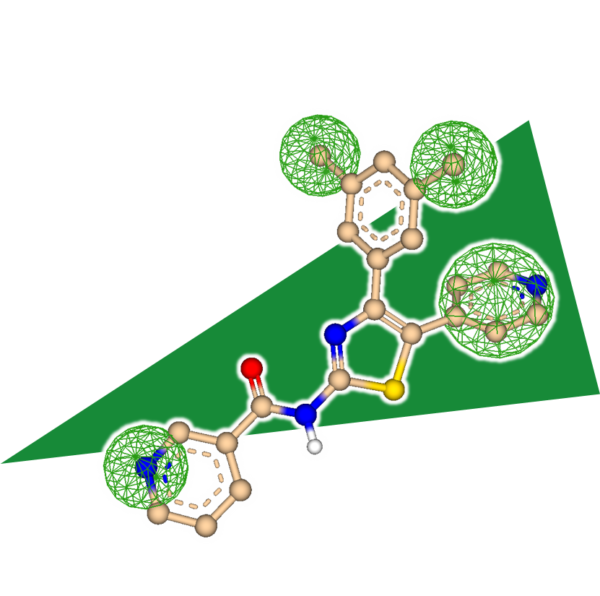
Explore the Beauty of Molecular Shape
After decades of structure elucidation, knowledge about the exact binding mode of a ligand remains a scientific luxury of the few. As a result, projects sometimes have to start from a single lead molecule without the possibility to rely on structural information about its interactions with the target. But how to proceed without knowing where to go? How to connect molecular features between unrelated scaffolds?
The answer: Embrace the beauty of what is already there.
SeeSAR 12, code-name 'Narcissus', enters the field of ligand-based drug discovery (LBDD) where the target's structure becomes an optional supplement.