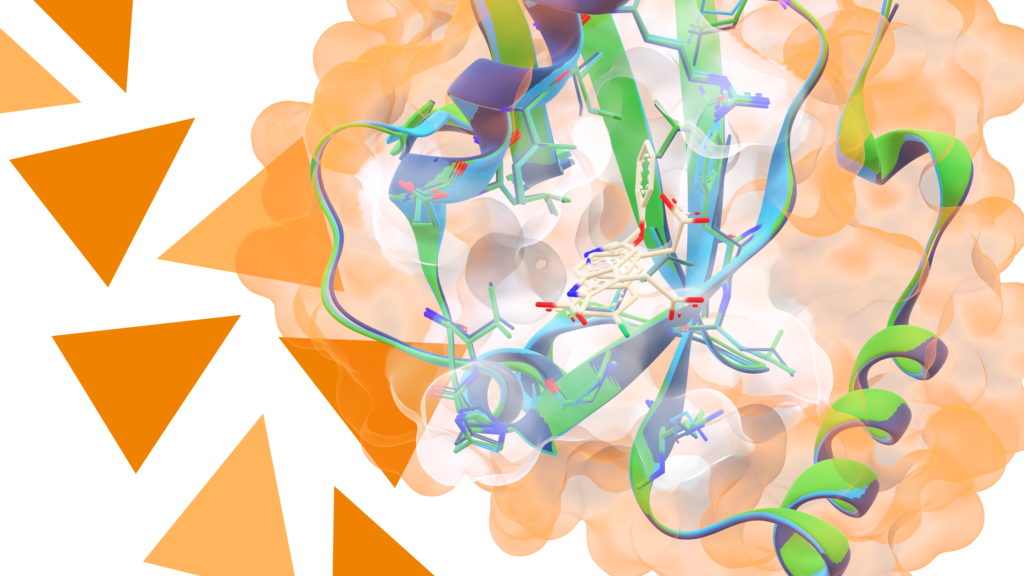
In the context of growing resistance of bacteria against antibiotics, the development of novel antimicrobial drugs and concepts against bacterial pathogens is of vast importance. In particular, Gram-negative pathogens pose big challenges to drug development due to the effective permeation barrier of the Gram-negative cell envelope. A smart concept for the development of novel antimicrobials is based on the conjugation of antimicrobial drugs with so-called “siderophores”, small molecule iron(III)-chelators, as carrier molecules: They show an active uptake mechanism for translocation over the cell envelope barrier. In this context, analogs of enterobactin, a tris-catecholate siderophore, play an important role during host infection and are of high interest.
Philipp’s team utilized a γ-modified L-allo threonine derivative as building block for the synthesis of biomimetic enterobactin analogs; they exhibit an attachment point for drug or reporter conjugation in the backbone of the siderophore — and simultaneously retain the hydrolyzability of the tris-lactone structure, that is important for entering the cytosol. Based on this approach the Klahn lab synthesized novel siderophore-fluororophore conjugates and drug conjugates. Philipp will take you through this exciting journey and is looking forward to a vivid discussion.